Top-Down for the Masses
Overview
Strong arguments exist for utilizing intact proteins as the primary unit of measurement in mass spectrometry-based proteomics. The proteoform hypothesis, which suggests that it is specific proteoforms that are most tightly linked to phenotype, resonates strongly with researchers performing both translational and basic science. In the previous granting period, the NRTDP developed potent quantitative workflows for discovery science and applied these workflows to myriad biological systems. Now, we have chosen to create an entire TR&D focused on making our quantitative workflows robust enough that they can be exported to non-expert labs through Community Engagement. To accomplish this, each step in these workflows, from sample preparation and separation to mass spectrometric detection and data analysis will be innovated to improve robustness. New sample preparation technology coupled with more robust, higher flow chromatography will alleviate issues related to nanoflow chromatography. Cleaner samples coupled with field asymmetric ion mobility spectrometry (FAIMS) will also improve mass spectrometer up-time. In addition to improved hardware robustness, the resource will also publish updated software tools, including AUTOPILOT ACQUIRE, TDPORTAL and TDVIEWER, which include streamlined quantitative data acquisition, processing, and analysis workflows. The combination of these innovations will give the resource the capability to efficiently export the experiments developed in-house to a community that has a growing appetite for performing experiments that are proteoform-resolved.
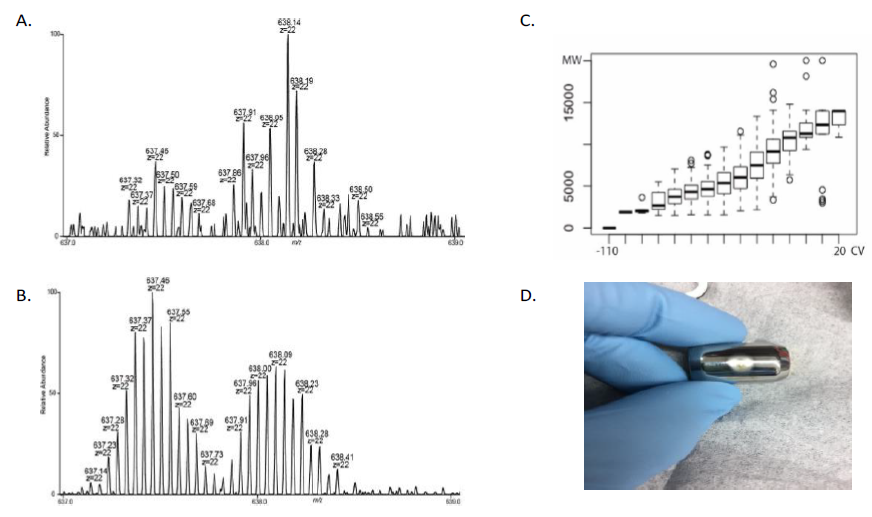
Field Asymmetric Ion Mobility Spectrometry (FAIMS)
Ion sources built into current mass spectrometers are often developed for the analysis of small molecules and peptides. When proteins and small molecules are co-analyzed in an electrospray-based system, the small molecules are often preferentially ionized. Mass spectrometers used for top-down proteomics also require frequent instrument cleaning due to deposition of neutral molecules onto ion optics. In this TR&D, we will use FAIMS to ameliorate both of these issues.


Comments are closed.